Anti-Aging: State of the Art
Aging is a problem that ought to be solved, and most Less Wrongers recognize this. However, few members of the community seem to be aware of the current state of the anti-aging field, and how close we are to developing effective anti-aging therapies. As a result, there is a much greater (and in my opinion, irrational) overemphasis on the Plan B of cryonics for life extension, rather than Plan A of solving aging. Both are important, but the latter is under-emphasised despite being a potentially more feasible strategy for life extension given the potentially high probability that cryonics will not work.
Today, there are over 130 longevity biotechnology companies and over 50 anti-aging drugs in clinical trials in humans. The evidence is promising that in the next 5-10 years, we will start seeing robust evidence that aging can be therapeutically slowed or reversed in humans. Whether we live to see anti-aging therapies to keep us alive indefinitely (i.e. whether we make it to longevity escape velocity) depends on how much traction and funding the field gets in coming decades.
In this post, I summarise the state of the art of the anti-aging field (also known as longevity biotechnology, rejuvenation biotechnology, translational biogerontology or geroscience). If you feel you already possess the necessary background on aging, feel free to skip to Part V.
Part I: Why is Aging a problem?
Aging is the biggest killer worldwide, and also the largest source of morbidity. Aging kills 100,000 people per day; more than twice the sum of all other causes of death. This equates to 37 million people—a population the size of Canada—dying per year of aging. In developed countries, 9 out of 10 deaths are due to aging.
Aging also accounts for more than 30% of all disability-adjusted life years lost (DALYs); more than any other single cause. Deaths due to aging are not usually quick and painless, but preceded by 10-15 years of chronic illnesses such as cancer, type 2 diabetes and Alzheimer’s disease. Quality of life typically deteriorates in older age, and the highest rates of depression worldwide are among the elderly.
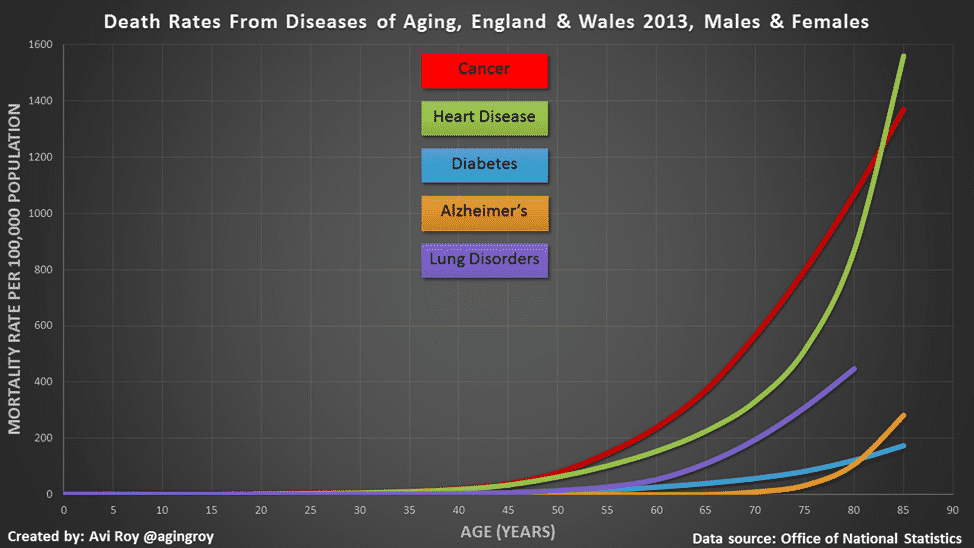
To give a relevant example of the effects of aging, consider that aging is primarily responsible for almost all COVID-19 deaths. This is observable in the strong association of COVID-19 mortality with age (below, middle panel):
The death rate from COVID-19 increases exponentially with age (above, middle). This is not a coincidence—it is because biological aging weakens the immune system and results in a much higher chance of death from COVID-19. On a side note, waning immunity with age also increases cancer risk, as another example of how aging is associated with chronic illness.
The mortality rate doubling time for COVID-19 is close to the all-cause mortality rate doubling time, suggesting that people who die of COVID-19 are really dying of aging. Without aging, COVID-19 would not be a global pandemic, since the death rate in individuals below 30 years old is extremely low.
Part II: What does a world without aging look like?
For those who have broken free of the pro-aging trance and recognise aging as a problem, there is the further challenge of imagining a world without aging. The prominent ‘black mirror’ portrayals of immortality as a curse or hubristic may distort our model of what a world with anti-aging actually looks like.
The ‘white mirror’ of aging is a world in which biological age is halted at 20-30 years, and people maintain optimal health for a much longer or indefinite period of time. Although people will still age chronologically (exist over time) they will not undergo physical and cognitive decline associated with biological aging. At chronological ages of 70s, 80s, even 200s, they would maintain the physical appearance and much lower disease risk of a 20-30-year-old.
This may sound like science fiction but is a phenomenon exhibited by other species such as hydras, naked mole rats, tortoises, whales, and sharks—the latter of which can live up to 400 years old. While these species do eventually die, their risk of disease does not change over time—a phenomenon known as ‘negligible senescence’ - and these species do not age. In contrast, as humans, we experience an exponentially increasing risk of death over time due to aging, a phenomenon known as Gompertz law. Yet this law is not an ingrained law of biology or the result of entropy, as it does not apply to other species, and the goal of anti-aging is to attain negligible senescence in humans.
There would be many benefits to an ageless population such as:
Very low rates of cancer, heart disease, Alzheimer’s disease etc.
Increased healthy lifespans
Increased cognitive function in older age
Lower death rates globally
Trillions of dollars saved on healthcare systems globally
Transitioning to an ageless population would come with several social implications that will need to be considered such as overpopulation, climate impact, immortal dictators and distributional justice. I’ll save a deeper discussion of these for a future post, but you can read responses to these objections here and by Aubrey de Grey, David Wood and others.
Part III: What is aging?
Aging is essentially damage that accumulates over time, which exponentially increases the risk of the diseases that kill most people (shown below):
This ‘damage’ associated with aging comes in essentially 9 forms, known as the hallmarks of aging:
Genomic instability
Telomere attrition
Epigenetic alterations
Loss of proteostasis
Deregulated nutrient-sensing
Mitochondrial dysfunction
Cellular senescence
Stem cell exhaustion
Altered intercellular communication
The hallmarks of aging are shown in the context of the cellular and extracellular microenvironment are depicted below:
These forms of cellular damage drive the increased risk of disease, frailty, cognitive decline as well as observable signs of aging such as grey hair, frailty and wrinkles. I’m going to save a deeper discussion of the hallmarks and their link to chronic diseases for a future post, but for excellent reviews on this topic I recommend this, this and this.
Where does this ‘damage’ come from?
The ‘damage’ (hallmarks of aging) occurs as a by-product of normal metabolism—the biochemical reactions that keep us alive. More and more damage accumulates and eventually leads to pathology, i.e. disease. When we talk about anti-aging we are talking about fixing the damage using an engineering approach before it accumulates to a dangerous level at which diseases emerge.
This basic model of aging can be understood as similar to the damage accumulated by a car. In its normal use, a car accrues damage that increases the likelihood that it will break down. Anti-aging is equivalent to maintaining a car, to prevent it from breaking down in the first place.
Anti-aging vs current medicine
Anti-aging is more feasible for extending healthy lifespan rather than solving the individual diseases of aging due to Taueber’s paradox and the highly comorbid nature of age-related diseases. Even if a person survives one age-related disease such as cancer, another (e.g. diabetes, cardiovascular disease) will kill them if aging is not solved. This accounts for the much smaller increase in healthy lifespan associated with curing the diseases of aging, such as cancer (2-3 years), versus slowing aging itself (30+ years):
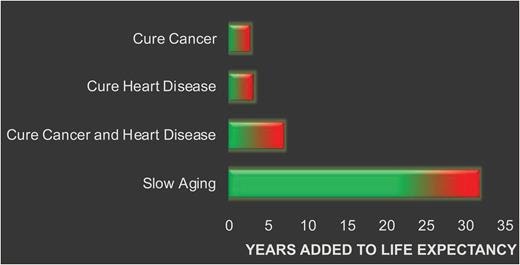
The difference between anti-aging and current medicine is the former prevents illness by targeting the hallmarks of aging, whereas the latter intervenes once a disease has emerged. If we compare current medical interventions associated with geriatrics with anti-aging—the former extends unhealthy lifespan, whereas only the latter extends healthy lifespan.
Therefore, there is strong reason to think that anti-aging will be more successful in extending healthy lifespan than the ‘sick-care’ approach of current medicine.
Part IV: Can aging actually be slowed?
In the lab, we have demonstrated that various anti-aging approaches can extend healthy lifespan in many model organisms including yeast, worms, fish, flies, mice and rats. Life extension of model organisms using anti-aging approaches ranges from 30% to 1000%:
These results demonstrate that aging is plastic, and not a fixed process. In mice, some of the most effective approaches to life extension are summarised below:
The plasticity of aging in model organisms that share similar metabolic physiology to us provides us good proof-of-principle that aging can be slowed in humans. It remains to be seen how much life extension is possible, and improved biomarkers of aging will be needed to accurately measure the effectiveness of new therapies in a reasonable time-frame.
Part V: Most promising anti-aging strategies
The past 5 years of research have demonstrated several anti-aging strategies as particularly promising. The diagram below, taken from a 2019 review by researchers at Stanford University summarises four of the most promising approaches to slow or reverse aging in humans, based on studies in mice:
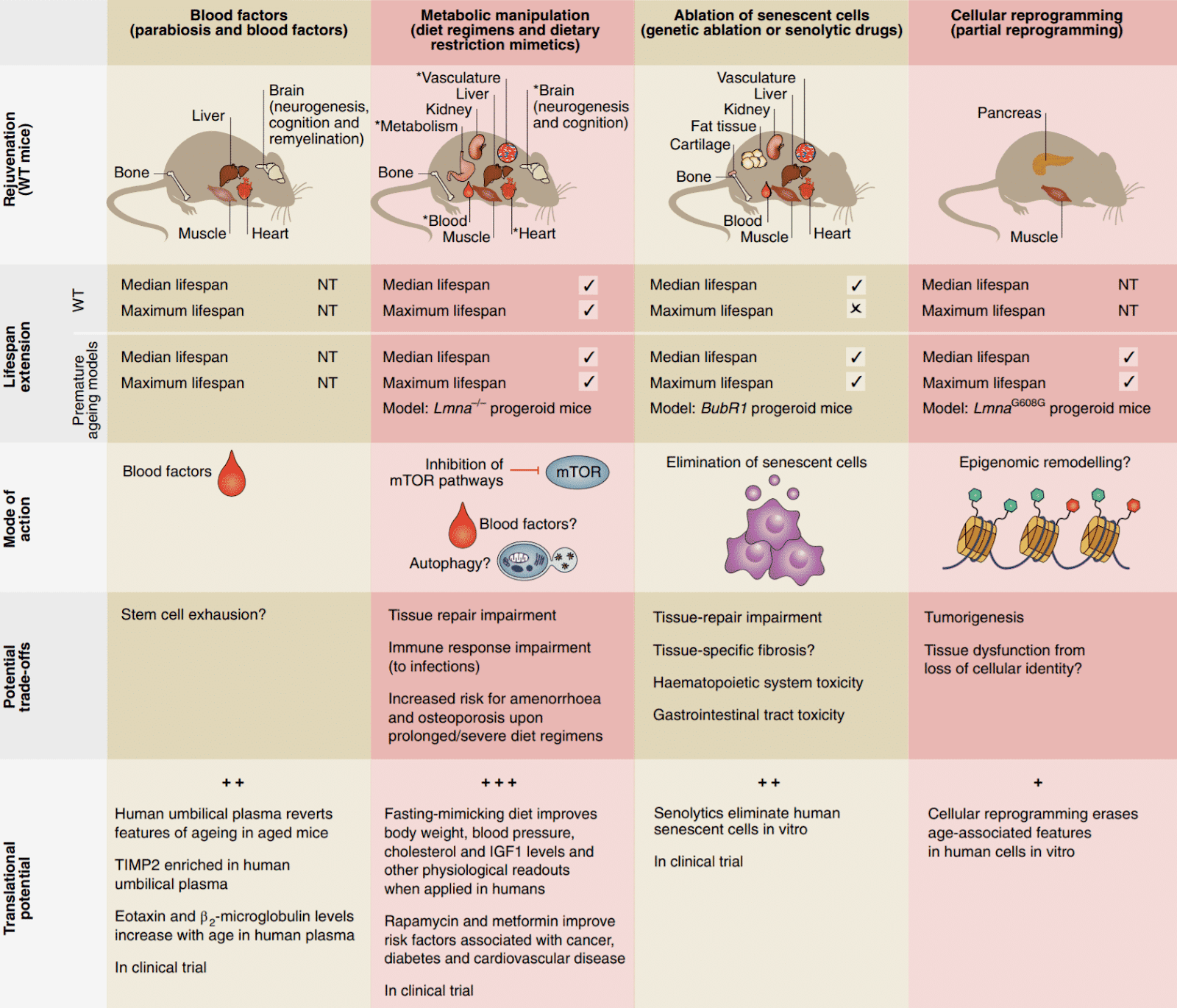
The above diagram may be quite technical for non-biomedical scientists, so I’ll briefly describe the approaches individually in simpler terms. Note that each of these strategies helps to reverse one or more of the hallmarks of aging.
1. Parabiosis (blood exchange)
Parabiosis (heterochronic parabiosis) is putting young blood into old mice, to make the old mice biologically younger. This is achieved in the lab by connecting the circulatory systems of young mice and old mice. Certain factors in the blood help to rejuvenate muscle, heart brain and liver tissues in old mice and restore their biological function.
Equivalent procedures that modify the compounds within blood in humans such as apheresis (blood filtering) could be used to slow aging in humans and thereby prevent or slow the progression of many types of age-related diseases including Alzheimer’s disease.
Recently, a group of Russian biohackers recently took part in the first plasma dilution experiments in humans. In a research context, the safety and effectiveness of apheresis is being tested in a clinical trial in humans by the company Alkahest.
Hallmarks reversed: parabiosis reverses age-related decline by targeting several hallmarks of aging including stem cell exhaustion, cellular senescence and altered intercellular communication (inflammation).
2. Metabolic manipulation (mTOR inhibitors)
Dietary restriction has been shown to extend healthy lifespan across several species. Drugs that mimic the metabolic effects of dietary restriction also have beneficial effects on lifespan. Nutrient-sensing biochemical pathways (such as IGF-1, mTOR and AMPK) play a key role in these effects. Metformin is a drug that is FDA-approved for diabetes that extends healthy lifespan in mice by inhibiting mTOR and activating autophagy. Metformin is currently being tested in a large clinical trial in humans to test its anti-aging properties.
Hallmarks of aging targeted: The widespread mechanisms of action of metformin help to improve all of the 9 hallmarks of aging, shown below. I’ll save the details for those interested, who can read a more thorough review here.
Another promising drug that manipulates metabolism is rapamycin (also known as siromilus), an FDA-approved immunosuppressant that extends healthy lifespan in mice and similarly acts to inhibit mTOR. Rapamycin is currently in a clinical trial in humans to test its anti-aging properties.
3. Senolytics—drugs that kill senescent cells
Senescent cells are a kind of ‘zombie’-like cell that accumulate with age. They are death-resistant cells that secrete proinflammatory factors associated with a range of age-related diseases (below, right):
There are various strategies being explored to kill or reprogram senescent cells (above, left), including senolytics. Senolytics are drugs that kill senescent cells to improve physical function and healthy lifespan. When administered to older mice, senolytics have been shown to reverse many aspects of aging such as cataracts, and arthritis (below):
Killing senescent cells with senolytics extends the median healthy lifespan by up to 27% in mice (below). Several senolytics, such as the combination of dasatinib and quercetin, and fisetin are in clinical trials in humans today.
Hallmarks of aging reversed: senolytics decelerate cellular senescence, improve epigenetic markers and restore intercellular communication (by reducing inflammation associated with senescent cells) to extend healthy lifespan.
4. Cellular reprogramming
Cellular reprogramming is the conversion of terminally differentiated cells (old cells) into induced pluripotent stem cells (IPSCs) (‘young’ cells). Cells can be re-programmed to a youthful state using a cocktail of 4 factors known as Yamanaka factors, a finding for which a Nobel prize was awarded in 2012.
Induced pluripotent stem cells (IPSCs) have essentially unlimited regenerative capacity and carry the promise for tissue replacement to counter age-related decline. Partial reprogramming in mice has shown promising results in alleviating age-related symptoms without increasing the risk of cancer.
An impressive example of cellular reprogramming was the restoration of vision in blind mice with a severed optic nerve using 3 of the 4 Yamanaka factors. The researchers from Harvard Medical School were able to regrow a fully functioning optic nerve in mice using cellular reprogramming. This approach could be used in future to regenerate other tissues as a new anti-aging strategy.
Hallmarks of aging targeted: Cellular reprogramming has been shown to reverse many of the hallmarks of aging, such as mitochondrial dysfunction, shortening of telomere length, changes in epigenetic marks, genomic instability, and cellular senescence.
5. Additional anti-aging approaches
Although not covered here, there are many other promising strategies for rejuvenation including thymic rejuvenation which has been shown to reverse biological age in humans, sirtuin enzyme activation with drugs such as resveratrol, and boosting mitochondrial function with NAD+ precursor molecules. All of these show the potential to increase healthy lifespan by targeting the hallmarks of aging.
Part VI: Conclusion
Aging is essentially damage accumulation that occurs as a by-product of metabolism and causes the diseases that kill most people today. This damage comes in 9 forms, which are the hallmarks of aging. Many therapeutic strategies show great promise in extending healthy human lifespan by reversing the damage accumulated with aging. Four of the most promising strategies to extend lifespan in humans include parabiosis, metabolic manipulation, senolytics, and cellular reprogramming.
Part VII: Call to Action
For those wanting to help aging be solved in our lifetime so we can avoid being the last generation to die, consider taking the following actions:
Sharing this post with others
Joining the Longevity subreddit or the Lifespan Discord server to plug into longevity channels
Learning more about the field (suggested reading materials below)
Donating to the SENS research foundation. They fund the most neglected and high-impact research in this field as explained in this LessWrong post.
Becoming involved in the longevity field as a researcher in one of the hundreds of laboratories around the world, or working for a longevity biotech company. You can find available jobs at LongevityList.com
Part VIII: Learn more
Books:
Ending Aging (2007) - Aubrey de Grey, PhD
Lifespan (2019) - Prof. David Sinclair, PhD
Age Later (2020) - Dr Nir Barzilai, PhD
Ageless (2020) - Andrew Steele, PhD
The Abolition of Aging (2016) - David Wood
YouTube:
Any talks by Prof David Sinclair, Dr Aubrey de Grey, Prof Brian Kennedy, or Dr Nir Barzilai for anti-aging science. For personal longevity strategies, I recommend talks by Dr Rhonda Patrick, Dave Asprey and Dr Peter Attia.
You can also follow the Oxford Society of Ageing and Longevity channel here.
Websites and blogs:
https://www.reddit.com/r/longevity
https://www.fightaging.org/
https://www.longevity.technology/
Good review articles:
Geroscience: linking aging to chronic disease (2014)
The business of anti-aging science (2017)
Turning back time with emerging rejuvenation strategies (2019)
From discoveries in aging research to therapeutics for healthy aging (2019)
If you wish to contact me outside this forum, please email me at jtt.harley@gmail.com
Edit:
Based on recommendations in the comments, I’ve added a short bio, below.
About me:
I’m not a geroscience researcher, but I am conducting neuroscience research at Oxford University after finishing my studies here and have a reasonable grasp of the geroscience field. I am vice president of the Oxford Society of Ageing and Longevity and in this role have had the privilege of interviewing influential figures in the field such as Aubrey de Grey, and researchers in the field such as Joao Pedro de Magalhaes. I’ve also had meetings with many other key figures in the field including David Sinclair. I have attended most of the major conferences in the field (ARDD2020, EARD2020, CSL Mechanisms of Aging 2020 etc.) last year, and read over 1000 academic papers in this field. I have also been been invited to give guest lectures on this topic at Oxford University and Monash University, Australia. I’ve been a content writer at Mindset Health for 2 years so I have some background in science communication.
- (The) Lightcone is nothing without its people: LW + Lighthaven’s big fundraiser by (30 Nov 2024 2:55 UTC; 612 points)
- Toss a bitcoin to your Lightcone – LW + Lighthaven’s 2026 fundraiser by (13 Dec 2025 19:32 UTC; 313 points)
- Book Launch: “The Carving of Reality,” Best of LessWrong vol. III by (16 Aug 2023 23:52 UTC; 131 points)
- The Fountain of Health: a First Principles Guide to Rejuvenation by (7 Jan 2023 18:34 UTC; 115 points)
- Voting Results for the 2020 Review by (2 Feb 2022 18:37 UTC; 108 points)
- 2020 Review Article by (14 Jan 2022 4:58 UTC; 74 points)
- What currents of thought on LessWrong do you want to see distilled? by (8 Jan 2021 21:43 UTC; 48 points)
- Why the outside view suggests that longevity escape velocity is a long time away and cryonics is a much more feasible option for those alive today: signal-boosting a comment by Calm-Meet9916 on Reddit by (8 Jan 2021 17:20 UTC; 32 points)
- Anti-Aging and EA (Recorded Talk) by (EA Forum; 17 Aug 2021 2:49 UTC; 21 points)
- 's comment on Book Review Review (end of the bounty program) by (17 Oct 2021 18:39 UTC; 8 points)
- 's comment on Aging should be effective altruism’s 5th cause area by (EA Forum; 2 Dec 2023 10:19 UTC; 3 points)
- Transhumanism 4 - LW/ACX Meetup #240 (Wednesday, June 7th 2023) by (5 Jun 2023 17:58 UTC; 2 points)
- 's comment on Anatomy, diseases and aging damage by (31 May 2021 17:14 UTC; 2 points)
I read this post at the same time as reading Ascani 2019 and Ricón 2021 in an attempt to get clear about anti-aging research. Comparing these three texts against each other, I would classify Ascani 2019 as trying to figure out whether focusing on anti-aging research is a good idea, Ricón 2021 trying to give a gearsy overview of the field (objective unlocked: get Nintil posts cross-posted to LessWrong), and this text as showing what has already been accomplished.
In that regard it succeeds perfectly well: The structure of Part V is so clean I suspect that it sweeps a bunch of complexity and alternative approaches under the rug, and the results described seriously impressed me and some of the people I was reading this text with at the time (We can reverse arthritis and cataracts in mice‽ We can double their maximum lifespan‽). It is excellent science propaganda: Inspiring awe at what has been accomplished, desire to accomplish more, and hope that this is possible.
While the post shines in parts III, IV and V, I have some quibbles and complaints about the introduction, part I and part II. First, I disliked the jab against cryonics in the first paragraph without considering the costs and benefits, which rightly received some pushback in the comments (the strongest counter-observation being that barring some practical suggestions for slowing down aging right now, cryonics and anti-aging research occupy very different parts of the strategy for life-extension, and can be pursued in parallel). Part II disappointed me because it was pro-longevity advocacy under the veneer of a factual question: Has anybody actually tried to think through how a world without aging might actually look like, instead of re-treading the same pro-aging trance and anti-aging science arguments? That seems like a question that is both interesting and pretty relevant, even when you believe that ending aging is important enough that it should definitely be done, if just to prepare for weird second- and third-order effects.
(Part I felt like I was a choir being preached to, which isn’t that bad, but still…)
I really liked learning a bunch of new facts about aging (as for example the list of species that don’t age, that aging is responsible for 30% of lost DALYs, and distinction between gerontology, engineering and geriatrics). Factposts are underrated.
The comments on this post were often very good, and had some nice discussion about whether the advice in section VII was to be focused on.
I’ve been overly negative in this review, but overall I still like this post, and have voted a 4 on it (which I might change to a 1). The parts III-V are excellent, and I have only minor problems with the preceding parts. This is the kind of post I would give a science-interested skeptic of anti-aging research. I’d like to have this post in the review, because it represents something some part of the core to the LessWrong transhumanist aesthetic that often gets overlooked.