How to have Polygenically Screened Children
Polygenic screening is a method for modifying the traits of future children via embryo selection. If that sounds like gobbledygook, then think of it a bit like choosing stats for your baby.
That may sound amazing. It may sound like science fiction. It may even sound horribly dystopian. But whatever your feelings, it is in fact possible. And these benefits are available right now for a price that, while expensive, is within reach for most middle-class families.
On a more serious note, there is limited selection power available with today’s technologies, so you will not be able to have a baby Einstein unless you are already a Nobel laureate. But polygenic screening will allow you to decrease your child’s risk of common diseases by 10-60%, reduce their risk of mental disorders, and increase their IQ by somewhere between 3 and 10 points. Including the cost of IVF and testing, these benefits are available for between $30k-100k depending on the mother’s age, how strong of a benefit you want and what kinds of traits you want to select for.
There has been quite a bit of discussion of this topic on LessWrong and adjacent communities but very little concrete advice for would-be parents who are curious whether the benefits are worth the price, particularly for those who have no other reason to do IVF. The purpose of this post is to fill that gap by addressing costs, potential medical complications, choice of clinic, which labs are best, and how age and infertility diagnosis affect the expected benefits.
This is a long post and I expect most people will not want to read the whole thing. If this is you, please use the section selector in the sidebar to navigate to the section you are most interested in. You may want to simply skip to the section titled “The Benefits of Polygenic Embryo Screening”.
Background on IVF
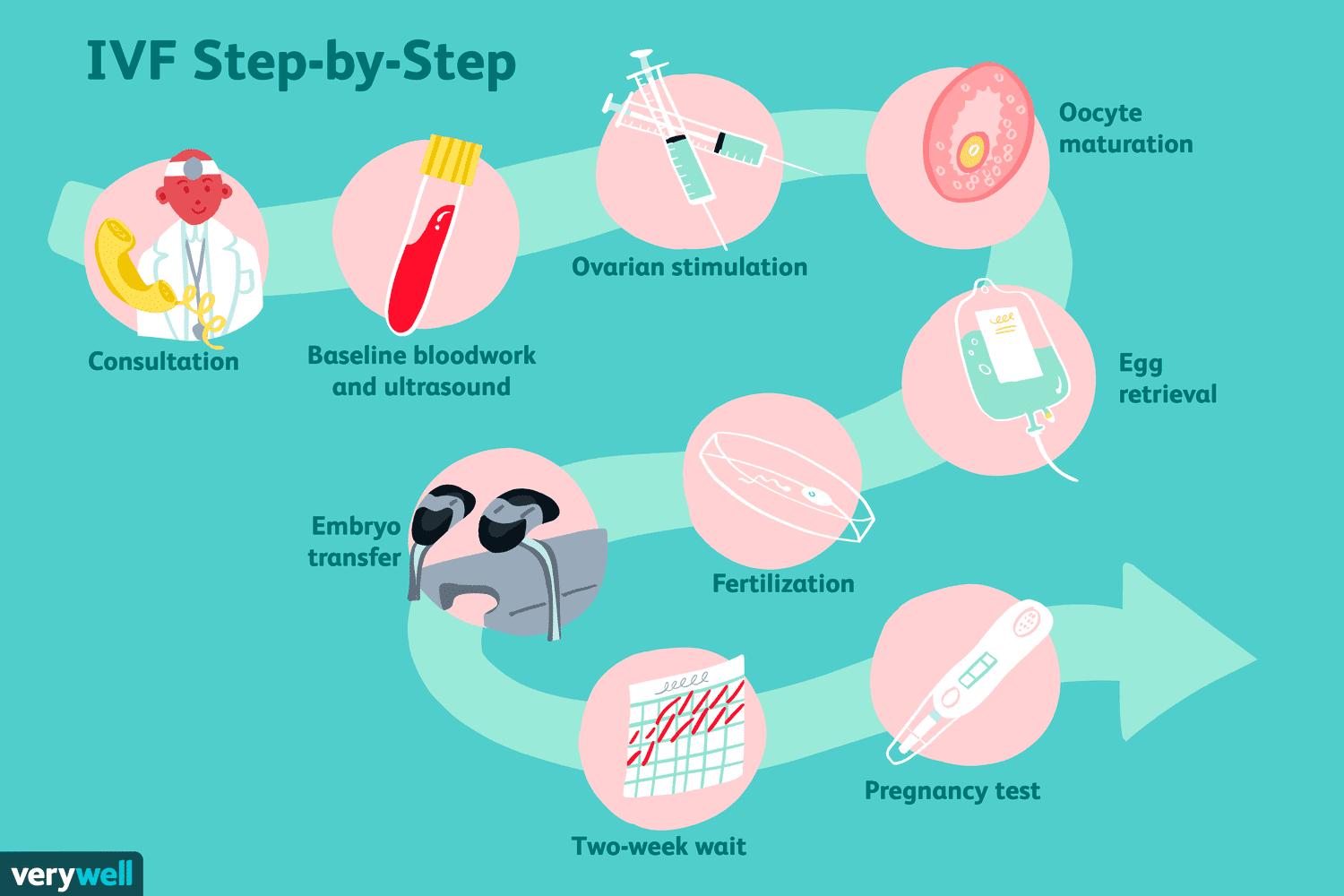
Wait, what even is polygenic embryo selection?
Embryo selection is all about picking an embryo to (hopefully) turn into a baby. This occurs during the process of In-Vitro Fertilization, or IVF. In the typical IVF cycle, a couple goes into a fertility clinic because they want to have a baby. Usually this is because they’ve been having trouble conceiving naturally, but couples also seek out IVF when they want to do genetic testing, select the sex of their child, or to preserve fertility for later pregnancy.
The doctor conducts a bunch of medical tests, and if they all check out, the woman begins a hormone regimen that will stimulate an abnormally large number of her eggs to mature all at once.
At the end of the regiment, the doctor extracts a bunch of mature eggs from the woman’s ovaries, which are then fertilized using the father’s sperm and grown in a lab dish for 4-7 days. When the embryo has finished growing, there are often four or more that can be implanted in the mother. Most couples do not want four children, so a choice must be made about which embryo to pick.
In ye olden days, doctors would often just transfer all the embryos at once in the hope that at least one of them would result in a baby. Sometimes this would work well; one of the embryos would happen to stick and the parents would be very happy. Other times it would work a little too well and more than one of the embryos would implant. This is why twin births are so much more common during IVF than during normal pregnancy.
Transferring multiple embryos at a time is less common nowadays because IVF clinics have figured out how to reduce the odds of failed pregnancy using genetic testing. With a higher chance of live birth from a single embryo transfer, the risk of a failed embryo transfer is outweighed by the risks of a twin pregnancy. The outcomes for twin births are on average worse than for single pregnancies. Twins are more likely to be born preterm, develop health problems, and put excess stress on the mother’s body.
This brings us back to embryo selection; the doctor or embryologist has to make a choice about which embryo to transfer first. All clinics have to make this choice, so all practice embryo selection of some kind. But the criteria for selecting the embryo have, until recently, been pretty dumb.
The standard practice is for an embryologist to look at all the embryos under a microscope and pick the one that looks the prettiest. I am not kidding. The embryologist will rank the embryos from best to worst based on their “morphology”, which accounts for factors like their rotational symmetry and whether or not they have a dark and rough colored appearance.
To be fair to the embryologists, this method is better than just randomly picking an embryo; embryos with particularly bad morphology gradings do actually have a lower chance of resulting in a live birth. And for a long time, there was simply no other option But times have changed and we can now select embryos by DNA rather than simply the appearance of their cells under a microscope.
But how do they even get an embryo’s DNA?
All the best techniques for genotyping rely on destructive sequencing, meaning the cells whose DNA is read must be destroyed. Embryos don’t have very many cells. So how do we get information about what’s in its genome without destroying it?
It turns out that after roughly five days of development, embryos possess a very interesting property; one may remove up to about 10 cells with little to no measurable impact on the embryo’s ability to develop into a healthy child. The embryo can regenerate up to about 10% of its mass! That’s the equivalent of losing and then regrowing both your arms as an adult. This is very fortunate for us because these cells contain a treasure trove of information.
The most common thing IVF clinics look for is aneuploidy, which is a medical term meaning “this embryo has an abnormal number of chromosomes”. The term for this type of testing is “PGT-A”, and it’s performed in roughly half of all IVF cycles in the US today.
Human embryos with the wrong number of chromosomes are surprisingly common, both among IVF patients and natural pregnancy. But this wasn’t very well understood before the first use of pre-implantation genetic testing in the late 1980s.
IVF Doctors started wondering why so many transfers were failing to result in pregnancy, or resulting in pregnancy followed by very early miscarriage. They discovered that roughly a third(!) of all pregnancies, both natural and via IVF have chromosomal abnormalities. Most of the time these go undetected because their immediate effect is to result in the arrest of the embryo’s growth, or to cause a very early miscarriage (often before the woman even knows she is pregnant).
IVF clinics began testing embryos for aneuploidy in the early 1980s. But in the late 2000s, something happened that completely changed the landscape of genetic testing.
Why Polygenic Screening wasn’t possible before 2015
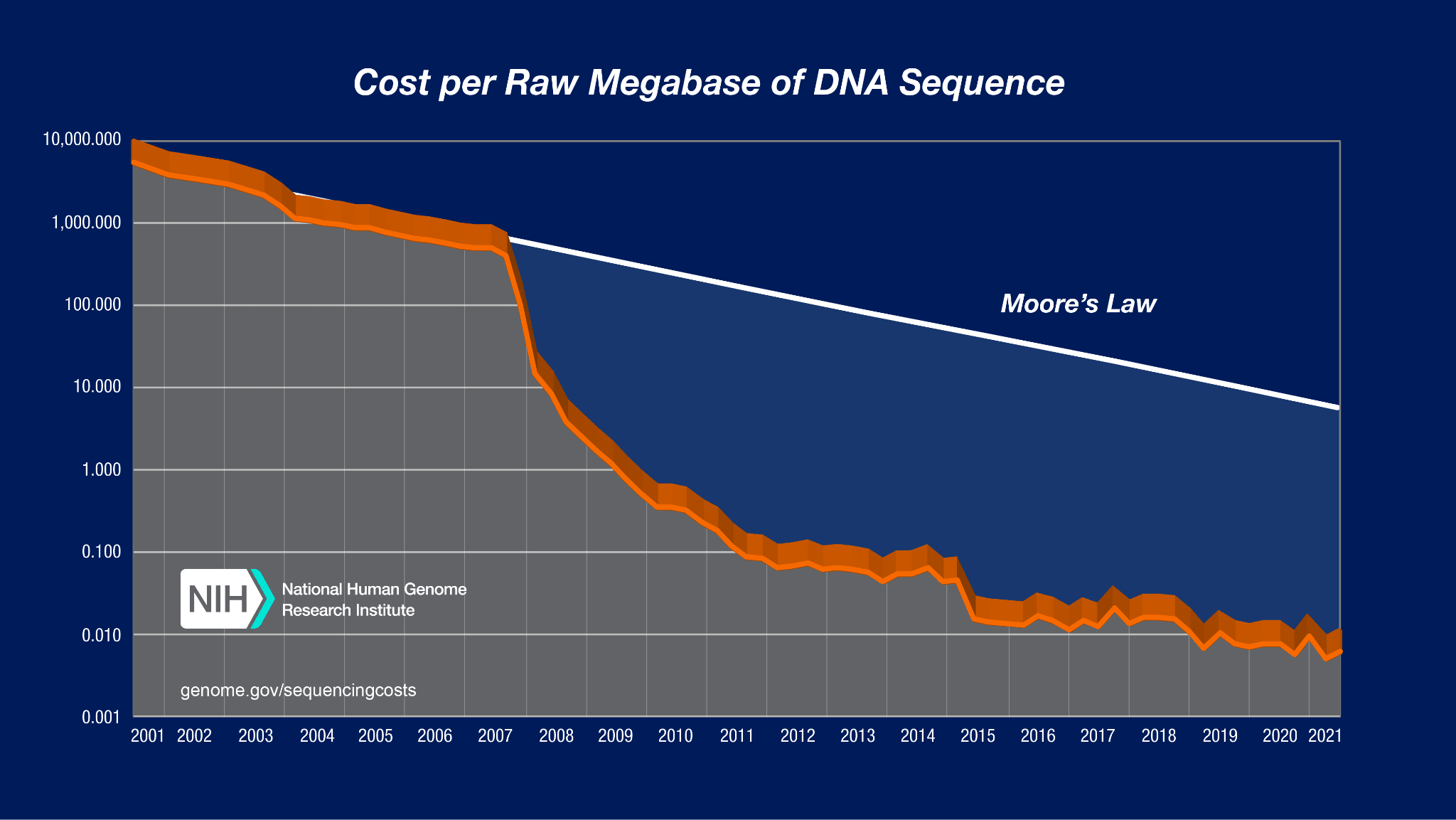
The cost of sequencing a fixed amount of DNA has declined dramatically since Fred Sanger and his team pioneered the first methods in 1977. There is a kind of “Moore’s law of sequencing” in which the cost of sequencing a fixed amount of DNA has declined exponentially over time.
However, something incredibly dramatic happened to DNA sequencing in about 2007. Take a look at this graph:
I don’t think I’ve ever seen a graph that looks like this anywhere else. Between 2007 and 2010, the cost of sequencing a megabase of DNA dropped by a factor of a million! That unbelievable, super exponential drop was made possible by a technology called “Next Generation Sequencing”.
By the mid-2010s, you could genotype all the parts of a person’s DNA most likely to differ from other people’s for under $100. At that price point, it became possible to gather genomes from hundreds of thousands of people and assemble them into giant databases that researchers could access.
This was incredibly important, because you NEED hundreds of thousands of samples to make good genetic predictors. It turns out that most of the traits we care about like heart disease risk or intelligence or attractiveness are determined not by a handful of high-impact genes, but by the cumulative effect of thousands of genes, each of which has only a tiny impact.
Take educational attainment. Educational attainment is not the most heritable trait, but because research on it is more politically acceptable in a university environment than the direct study of intelligence, we know quite a bit about its genetic roots. The latest large-scale study of it included data from 2.7 million participants. Among all genes identified, the one with the single largest effect size only increased the amount of time you spent in school by at most 2.8 weeks (see section 3.4). That’s it! The average gene has a tiny, tiny impact on how long you spend in school. The predictor used 2,925 genes to explain just 15% of the variance in how many years of school a person completed.
So you actually NEED these gigantic databases to explain more than a tiny fraction of the variance in complex traits. This is why polygenic embryo selection was impossible before about 2016; there just wasn’t enough data to figure out which genes did what.
How do they know which genes do what?
Once you have a giant biobank and information about people’s traits and diseases, you still need to figure out which genes do what. I mentioned an educational attainment predictor in the section above, but I didn’t explain how they created it. So how did they do it?
The answer is actually not too complicated: a researcher will use one of these gigantic biobanks plus a machine learning model to identify which genetic variants are associated with an increase or decrease in a given trait.
The dumbest possible way to do this is with a Genome Wide Association Study, or GWAS. It works something like this:
Let’s say there’s a gene with two different variants commonly present in the biobank population. 96% of participants have an “A” at some particular location in the gene, but 4% have a “T” instead. We want to know whether having a “T” makes you taller.
A GWAS just measures the average height of people with an A and the height of people with a T to see if they’re different. Then it uses a statistical significance test to see if the result could have plausibly been the result of random chance. If not, the researchers reason that the gene is having an effect on height. If it passes this test, it is added to the “list of important genes for height”.
For such a dumb method, this works remarkably well. Height predictors created using GWAS results correlate with actual height at about 0.55.
The smarter way to do this is to use some kind of machine learning method like LASSO. This will give you a better predictor for the same amount of data. But for some reason I still don’t really understand, academia almost exclusively uses GWAS.
Correlation or causation?
“OK”, you might say. “That’s well and good, but how do we know that these genetic differences are actually CAUSING someone to be taller or smarter rather than just spuriously correlated with height?”
The main reason this is possible is because nature has already conducted a randomized control trial on our behalf. Every time your body produces a sperm or egg cell, your DNA is more or less randomly mixed up and half of it is given to the reproductive cell. This means that, conditional on parental genomes, sibling genomes are randomized!
In turn, this means that if a gene can predict differences between siblings, you can be quite confident that it is in fact CAUSING the difference. This is actually quite a remarkable fact, and one that underpins the entire reason for believing embryo selection should work.
There is one asterisk here; though a sibling GWAS can tell you where the causal variant is, it usually can only narrow down the list of candidates to perhaps 10 distinct variants within a region of very roughly 100,000 base pairs. This is sufficient for embryo selection because that set of 10 base pairs will almost always be inherited together. But if sometime down the line we want to do embryo editing, it will require us to either pinpoint the causal variant precisely or to edit all 10 variants that have a decent chance of causing the observed change.
Another crucial insight from these studies is that nearly all of the genetic differences between humans can be explained by additive effects; there are very few gene-gene interactions going on; If gene A makes you taller, it doesn’t depend on gene B being present to work its magic. It’s a strong, independent gene that don’t need no help.
This fact is extremely important because it makes both evolution and embryo selection possible. There is a common misconception that genes are tied together in a hopelessly complex web and that if we mess with one part of it the whole thing will come crashing down. While that may be true for genes that are universally present in the human population, it is very rarely true for genes that commonly vary between people.
You have the predictors. Now what?
Once you have created genetic predictors using GWAS or LASSO or some other method, you can then feed in the embryo’s DNA to the trained model and get a prediction of each embryo’s expected trait value. You do this for every predictor you have (or at least those you care about), and then pick an embryo based on the results.
But there’s one last question to answer: which traits should you care about? If one embryo has a 20% chance of getting breast cancer and a 10% chance of getting heart disease, is that better or worse than an embryo with a 10% risk of breast cancer and a 20% risk of heart disease? Or how about one that has a high risk of both but is also predicted to have an IQ 5 points above average?
There is no universally agreed-upon method for making the choice about which embryo to implant. My personal hope is that someone (maybe even me!) makes a tool to assess what parents find important and then ranks embryos according to those criteria.
The Benefits of Polygenic Embryo Screening
| Category | Trait | Improvement Range | Publicly Available? |
|---|---|---|---|
| Non-disease | Intelligence | +1.6-7.5 IQ points | Yes* |
| Non-disease | Height | 1-6 cm | Yes* |
| Non-disease | Personality | ¯\_(ツ)_/¯ | Yes* |
| Disease | Alzheimer’s | 15-50% reduction | Yes |
| Disease | Atrial Fibrillation | 10-50% | Yes |
| Disease | Asthma | 3-50% | Yes |
| Disease | Breast Cancer | 3-50% | Yes |
| Disease | Basal Cell Carcinoma | 3-45% | Yes |
| Disease | Coronary Artery Disease | 20-60% | Yes |
| Disease | Gout | 12-70% | Yes |
| Disease | Heart Attack | 25-70% | Yes |
| Disease | High Cholesterol | 12-50% | Yes |
| Disease | Hypertension | 10-45% | Yes |
| Disease | Inflammatory Bowel Disorder | 5-65% | Yes |
| Disease | Ischemic Stroke | 5-20% | Yes |
| Disease | Melanoma | 0-35% | Yes |
| Disease | Obesity | 12-65% | Yes |
| Disease | Prostate Cancer | 2-60% | Yes |
| Disease | Type 1 Diabetes | 10-55% | Yes |
| Disease | Type 2 Diabetes | 20-60% | Yes |
| Disease | Testicular Cancer | 0-55% | Yes |
| Mental Disorder | Major Depressive Disorder | 5-20% | Yes |
| Mental Disorder | Schizophrenia | 5-75% | Yes |
*See the section below for how to get access to these predictors
Ok, enough with the theory. How big of a benefit can you actually get from going through IVF and screening your embryos?
I’ll start with the one everyone always asks about: intelligence. How much can you boost your child’s IQ with embryo selection?
How much can embryo selection increase my child’s IQ?
First, there is no company that publicly offers embryo selection for intelligence. I have spoken with a stealth mode startup that offers selection for disease and non-disease traits, including intelligence. If you’re interested in selecting for non-disease triats, you can get in touch with them via Jonathan Anomaly, who knows some of the people working at the company.
Their current predictor correlates with measured IQ at about 0.4, which means they’ve likely compiled data from multiple sources to create it.
So how big would the gain be? Using some code from Gwern’s monster post on embryo selection for intelligence and some results from their calculator, I created the following graph:
It’s plausible that you would get up to maybe 40 euploid embryos if the mother is young and you do multiple rounds of egg retrieval. In that case, you could probably get a gain closer to 8 points. If the mother is older it will be less. There’s also a reduction in benefit if one of the parents is of non-european ancestry: likely around 8% for Ashkenazis, and 20% for east asians, and probably a similar or lesser reduction for Indians. I am uncertain of the reduction in gain for those of African ancestry, but it would likely be larger (perhaps 30-40%?).
This is an unfortunate side-effect of the fact that there aren’t enough non-Europeans in the large biobanks on which these predictors are trained.
There is significant room for this benefit to improve in the near future. The million veterans project in the US has whole genome sequences and ASVAB test scores for (you guessed it), a million soldiers. If researchers were simply allowed to use this existing data to create an intelligence predictor, the gain from embryo selectioin would increase to 8.5-13 IQ points and the racial disparities in predictor quality would mostly disappear. The marginal cost of this would be virtually zero.
This future increase in the efficacy of embryo selection has an obvious implication: if you freeze eggs or embryos now, you’ll have more embryos to pick from (since egg production declines with maternal age), and if you wait a few years to implant them, the expected gain from selection will be higher.
Disease Reduction
Unlike intelligence, there actually are several companies that offer polygenic embryo screening for disease risk. For this reason, I can tell you quite a bit about exactly how much you can reduce disease via embryo selection.
There are two main “categories” of disease risk to think about: monogenic disease risk (which includes diseases like Tay Sachs, Cystic Fibrosis etc), and polygenic disease risk, which includes heart disease, alzheimers, schizophrenia, diabetes and most others.
I’m going to focus on polygenic screening, since everyone has a non-zero risk of them, and embryo selection to reduce polygenic disease risk is already available in clinics.
The first company to offer this was Genomic Prediction. Orchid Health also finally offers polygenic embryo screening, though I believe they charge more per embryo. One other lab in China appears to have recently deployed a similar test, though it looks as though they screened embryos for type 2 diabetes risk exclusively, which is not a very sensible strategy in my opinion. And lastly, there is a stealth mode startup doing polygenic embryo screening. They offer screening for both disease risks and non-disease traits like intelligence. If you’re interested in contacting them you can reach them through Jonathan Anomaly.
Because I have little information about the predictor quality of Orchid Health or the stealth mode startup, I will focus on papers published by Genomic Prediction.
They use a pretty straightforward and simple method for determining an embryo’s relative ranking: each disease is weighted according to its impact on disability-adjusted lifespan. According to one of their recent papers, selecting embryos in this manner results in a fairly impressive reduction in disease risk across multiple conditions. Here’s a graph from one of their latest papers showing the expected reduction in the relative risk of various diseases from selecting the best of five embryos.
The figure above shows disease reductions for selection among 5 “pseudosiblings” of European descent (apparently there are not enough real siblings to train predictors on). My guess is the benefits will be reduced compared to those shown, perhaps by around 20%.
Note also that this doesn’t take losses from implantation or miscarriage into account. So you’ll need more like 7 or 8 euploid embryos to achieve gains 20% lower than this. But still, the benefits are reasonably strong.
The most amazing thing to me about the above graph is that selection seems to reduce EVERY disease in the index. One of the major concerns I hear raised about embryo selection is that there might be some “hidden downside” to selection. This graph seems to suggest that, at least so far as diseases go, that’s not much of a concern.
Another way of looking at the benefits of disease reduction is to look at how much the quality-adjusted lifespan of an average child born via polygenic screening would increase compared to one born without its benefits. Here’s their analysis of this framing:
Keep in mind, this is the pseudosibling benefit, so take what you see on the graph above and multiply it by 0.8 to get a more realistic number. Also, the benefit is not quite as large for non-European groups. It looks like the gain is reduced by about 20-30% for South Asians, 30% for Africans and 35% for East Asians.
Of course, this all kind of ignores the elephant in the room: many of these diseases have an average age of onset of 50-75. In fifty years the world is likely to look incredibly different. If humans are still around, it seems likely that we’ll have cures or at least very effective treatments for many of these conditions.
The exceptions are mental disorders like clinical depression and ADHD, which generally have an average age of onset before age 25, and obesity, which is now showing up more and more in childhood.
If Orchid Health is more expensive, why would anyone use their tests?
Orchid Health offers whole genome embryo sequencing. That means instead of just looking at ~800,000 spots in the genome, they look at about 3 billion.
This doesn’t make as big of a difference as you might think; most of the information we care about is already present in one of those 800,000 spots that companies like Genomic Prediction examine with their embryo testing. But whole genome sequencing has one specific advantage that companies like Genomic Prediction can’t replicate: they can detect de novo mutations.
De novo mutations are random genetic mutations that aren’t present in either of the parents. When you think of “random mutation and natural selection”, the “random mutation” part IS de novo variants.
We hear a lot about mutations conferring evolutionary advantages, both in science class when learning about evolution and in science fiction stories like X-men, but unfortunately most de novo mutations are just bad for you. Some of them are REALLY bad, as in your child will die quickly after birth or live a permanently stunted life.
Orchid’s testing finds one of these monogenic issues in about 3.7% of embryos. I’ll update this post later with more concrete info about the size of the benefit from having a child without those problems, but at a high level I think it’s likely worth the price if you can afford it.
However, the price is very, very high. Orchid charges $2500/embryo. I believe Genomic Prediction is about $500/embryo.
Other Non-Disease Traits
Personality Traits
As with intelligence, there are no companies publicly offering screening for personality traits to the best of my knowledge. The stealth mode company has told me they may offer personality prediction at some point, though I am uncertain of their quality.
Predictors for personality traits such as conscientiousness and neuroticism are poor. This seems to be partly related to the lack of good data on such traits. A paper published in March of 2022 was able to explain 2% of the variance in conscientiousness and much less for others. There is significant room for improvement here, as most estimates peg the heritability of the big five personality traits at roughly 40-60%.
There are existing third-party services like Genomelink and SelfDecode which provide personality predictors. Unfortunately, they don’t have any published data on the quality of the predictors they use, and the anecdotes I’ve heard suggest that, at least for intelligence predictors, they are very poor. So if you’re looking for something that isn’t terrible, your best bet is to contact Jonathan Anomaly and hope the stealth mode startup offers something better.
Facial Features/Attractiveness
It would be nice (or possibly dystopian, depending on your views) if you could add physical attractiveness to your embryo selection criteria. More attractive people have higher lifetime income, better dating prospects, and seem to benefit generally from the halo effect.
To the best of my knowledge, no one offers this as a service right now, so unless the super secret startup offers it, you’d probably have to pay a researcher tens of thousands of dollars to develop a predictor for you.
The predictor might be halfway decent, though I don’t expect it to be as good as the ones we have for intelligence or most diseases. Here’s a meta-analysis of facial feature genetics from 2020 that found 203 genome-wide significant signals. If someone made a predictor using that paper it might be able to predict facial attractiveness reasonably well. However, there is clearly much room for improvement.
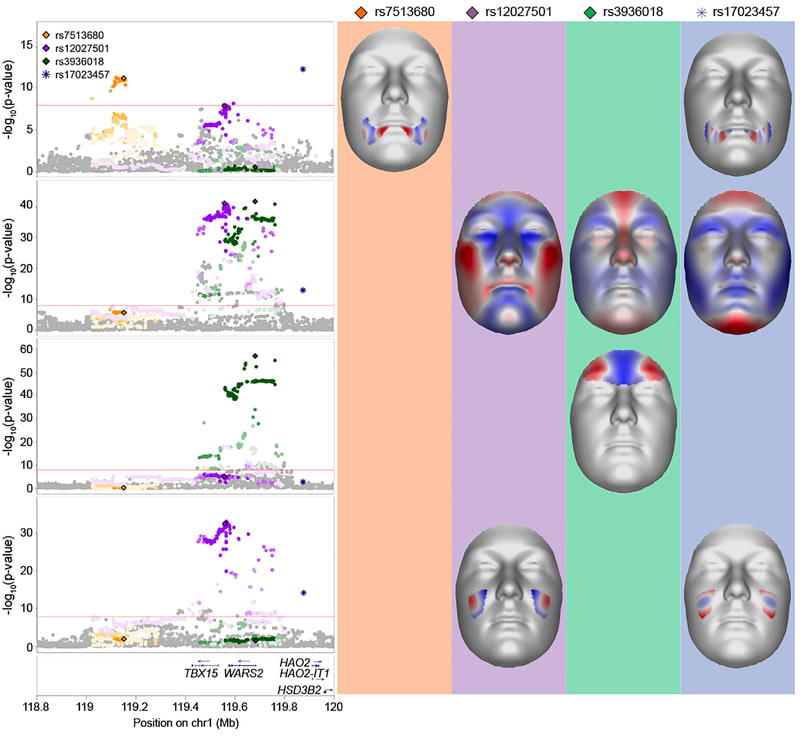
Is attractiveness a purely position good? That is to say, if everyone was made attractice, would society be better off? My guess is probably a little, but I haven’t done enough research into this topic to be sure.
If you want to see what selection on attractiveness looks like in the extreme, take a look at male birds of paradise. Female birds have been selecting mates based on their plumage and dancing ability for thousands of generations, resulting in some very beautiful, but very strange looking birds.

It is worth asking ourselves if dozens of generations of selection for attractiveness might result in similarly strange and pointless features whose sole purpose is to elicit arousal in members of the opposite sex.
And in the shorter run, selection for physical attractiveness probably trades off at least somewhat against selection for other features that have a greater benefit for society as a whole. So for now there is a non-zero cost to making everyone more attractive.
Still, from a purely selfish perspective, some level of selection for attractiveness makes sense. As with intelligence, no company currently offers the ability to select for physical attractiveness, unless you include selection for height, which already works extremely well. Your best bet is contacting Jonathan Anomaly to see if the groups he knows offer something. Or, barring that, you could hire a PHD student for 50k and pay them to develop a predictor for you.
Concrete Advice for Would-Be Parents
If you’ve read the sections above and you decided you want to do polygenic embryo screening, your primary objective should be to get maximum gain for minimum cost. This section is about how to achieve that goal.
There are two primary inputs that determine the effect size of polygenic screening:
The power of the polygenic predictors used to select an embryo for implantation.
The number of “achievable births”, meaning the number of children you would have if you implanted all of your embryos one by one.
Number 1 is largely out of your control unless your name is Gwern or you are a research scientist with access to a large biobank.
Number 2 IS within your control, at least to some degree. Here is a list of factors you might be able to change that will influence achievable births:
How many IVF cycles you go through
The IVF clinic you choose
The PGT lab used for aneuploidy testing
The age of the mother or egg donor at the time eggs are extracted (younger is always better)
The stimulation protocol used
The age of the father or sperm donor (younger is better)
Whether you choose to freeze eggs or embryos
Routine prenatal care that any good obstetrician will be able to tell you (though see Emily Oster’s excellent book if you want more details)
Why do these factors matter? Because at every step of the IVF process from initial consultation to birth, fewer eggs/embryos/pregnancies come out than go in. There’s a loss associated with each step, and the factors listed above have a large influence on the size of the loss. Here are some suggestions on how to reduce losses and costs:
Suggestion #1: Reduce uncertainty about how many euploid embryos you will produce
The single most important input into the “gain” equation is the number of mature eggs harvested per retrieval. However, this quantity has a very wide distribution. In conversations an acquaintance of mine had with egg donor clinics, they mentioned that some donors produce as many as 100 eggs per retrieval. A woman in her mid-40s with infertility issues might produce 3.
There are several heuristics you can use to reduce the uncertainty about how many eggs you are likely to harvest during an egg retrieval. Knowing these beforehand can help reduce uncertainty about the exact size of the benefits of polygenic screening.
The easiest heuristic to use is the woman’s age. As a general rule, I wouldn’t do IVF for the purpose of polygenic embryo selection unless the mother is under 38. Beyond age 38, the losses in the IVF funnel are just too steep to justify the pain and expense, unless you plan to use donor eggs.
Another lower-cost but not free thing you can do is assess your likely egg production by undergoing the very first part of IVF; a consultation and ovarian ultrasound. This ultrasound is performed right at the very start of the IVF process and usually costs less than $1000. Ovarian reserve and antral follicle count are strongly correlated with the expected number of mature eggs you or your female partner will produce after hormonal treatments. If you’re willing to do embryo selection in theory if the gain is large enough, this can be a relatively inexpensive way to reduce the uncertainty about the benefits of the procedure.
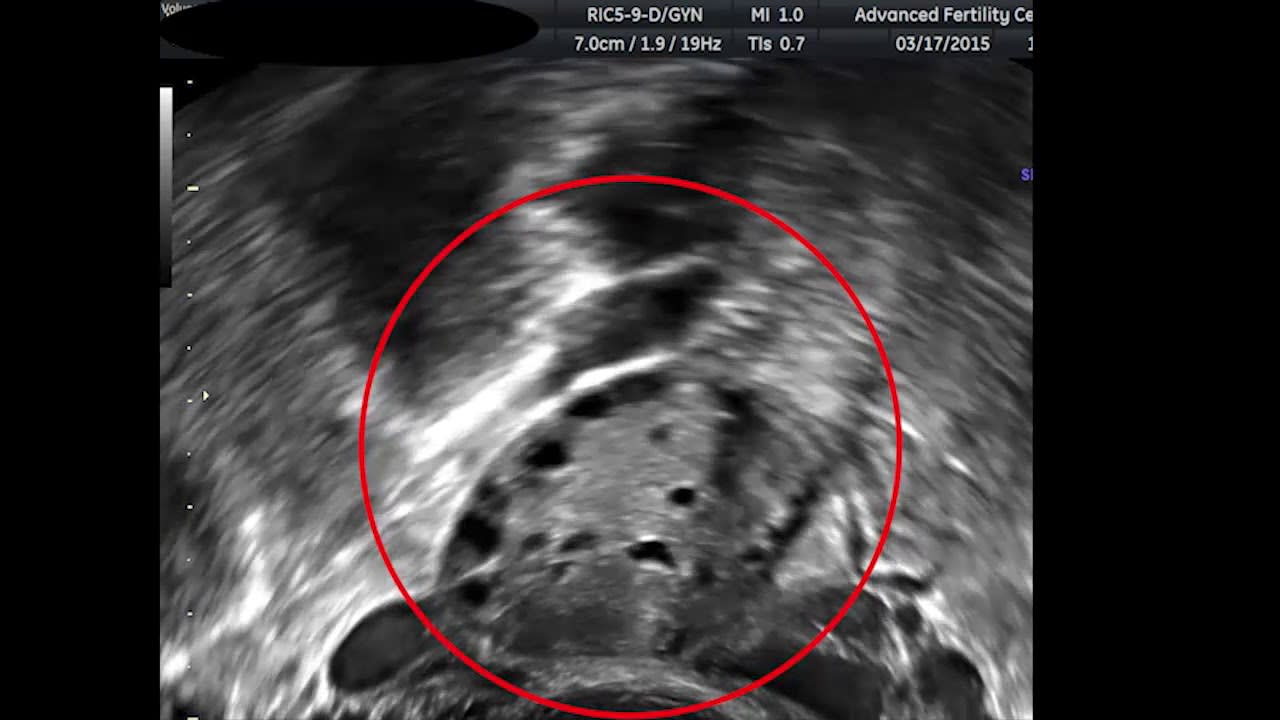
What is really needed here is a tool that allows one to see the expected gain across a variety of traits given an antral follicle count. The data necessary to do this research is present in the CDC’s NASS database, but I don’t yet have access. I hope to do this for a future research project. Until then, here’s a table showing roughly how your odds vary as a function of antral follicle count.
Suggestion #2: Use my table to pick a clinic
I have spent an embarrassing amount of time on a research project to rank every IVF clinic in the US from best to worst. I compiled this list using data from the CDC’s NASS database, which has information about clinics going back to the mid-90s. I believe I am the first and only person in the world to do this. If you’re curious about a clinic that isn’t on the last, feel free to reach out to me at genesmithlesswrong@gmail.com.
A somewhat boring explanation of the research I did (skip this if you just want results)

Clinics are ranked according to their cumulative live birth rate per intended egg retrieval among patients using their own eggs (not donor eggs). In simple English, that means we’re looking at what percentage of women who started hormone treatments actually delivered at least one child.
In an ideal world I’d give you clinic rankings based on the number of expected births per retrieval. But without access to intermediate outcome data from NASS, this is impossible. I plan to apply for access eventually, but in the meantime I think live birth rate is probably quite a good proxy, and clinics with very high live birth rates are likely to be able to produce more achievable births than those with lower live birth rates.
I’ve taken care to control for various factors that could confound the analysis. Some clinics attract a larger proportion of younger patients, who have better prospects than older cohorts. Some clinics attract a large proportion of patients solely interested in freezing eggs or embryos for some later treatment. Some clinics have a very small number of cycles each year and can score very well or very poorly depending on the luck of the draw. I’ve controlled for all of these confounders in my analysis.
The one big thing I didn’t control for was the percentage of patients presenting with a given infertility diagnosis. I attempted to to this in an earlier version of the project but had weeks and weeks of nightmares trying to find some defensible way to deal with the large amount of censored data present in spreadsheets. Supposedly the CDC censors these values to protect patient privacy. This justification is obviously nonsense; they still have uncensored values from before 2018 on their website. I emailed to ask if I could apply for some kind of special access as a researcher and was denied.
I tried to work around these missing vaues but eventually I simply gave up. I had to make too many unjustified assumptions to compute clinic rankings, and ranking was highly variable depending on which assumptions I made.
So this final analysis controls only for maternal age, use of patient’s eggs (vs donor eggs), percentage of retrievals conducted with the intention to freeze embryos or eggs (obviously those people aren’t going to have a baby), and some bayesian averaging sprinkled in on top to differentially bring clinics with low retrievals/year more towards the mean of all clinics.
I plan to actually publish my results in proper academic setting at some point, so this post contains only the headline numbers.
Without further ado, here are the top 25 IVF clinics in the US as of 2020.
The best IVF clinics in the USA
| Clinic Name | Adjusted Live Birth Rate | Clinic State | Phone Number |
| Carolinas Fertility Institute | 0.516123 | North Carolina | (336) 448-9100 |
| The Georgia Center for Reproductive Medicine (no website) | 0.490339 | Georgia | (912) 352-8588 |
| Reproductive Gynecology & Infertility-Westerville (Columbus location) | 0.482645 | Ohio | (614) 895-3333 |
| Reproductive Medicine Associates of New Jersey | 0.458511 | New Jersey | (973) 971-4600 |
| Center for Reproductive Medicine, Advanced Reproductive Technologies | 0.457538 | Minnesota | (612) 863-5390 |
| Missouri Fertility | 0.456779 | Missouri | (573) 443-4511 |
| Spring Fertility (San Francisco location) | 0.446066 | California | (415) 964-5618 |
| CCRM Boston (main center in Chestnut Hill) | 0.439929 | Massachusetts | (617) 449-9750 |
| SpringCreek Fertility (Dayton location) | 0.437458 | Ohio | (937) 458-5084 |
| Duke Fertility Center, Duke University Medical Center | 0.419579 | North Carolina | (919) 572-4673 |
| New Direction Fertility Centers | 0.413924 | Arizona | (480) 351-8222 |
| Shady Grove Fertility Colorado | 0.411165 | Colorado | (720) 704-8221 |
| Center for Advanced Reproductive Medicine | 0.409579 | Kansas | (913) 588-2229 |
| Fertility Center of the Carolinas | 0.406704 | South Carolina | (864) 455-1600 |
| Fertility Center of San Antonio | 0.403583 | Texas | (210) 692-0577 |
| Baystate Reproductive Medicine | 0.403039 | Massachusetts | (413) 794-1950 |
| University of Iowa Hospitals and Clinics, Center for Advanced Reproductive Care | 0.401587 | Iowa | (319) 356-8483 |
| Carilion Clinic Reproductive Medicine and Fertility | 0.400715 | Virginia | (540) 985-8078 |
| Advanced Fertility Center of Chicago | 0.399771 | Illinois | (847) 662-1818 |
| Shady Grove Fertility-Richmond | 0.394682 | Virginia | (804) 379-9000 |
| Fertility Center of Southern California | 0.391824 | California | (949) 955-0072 |
| Northern California Fertility Medical Center | 0.390555 | California | (916) 773-2229 |
| The Nevada Center for Reproductive Medicine | 0.390344 | Nevada | (775) 828-1200 |
| Center for Advanced Reproductive Services (Farmington Location) | 0.388567 | Connecticut | (844) 467-3483 |
For reference, the average adjusted live birth rate for all clinics nationwide was 0.278.
How predictive are an IVF clinic’s past success rates of their future success rates? Here’s a graph showing how well a clinic’s 2017 live birth rates correlated with their 2020 live birth rates after adjusting for the confounders I mentioned above:
The overall takeaway here is fairly clear: by selecting one of the top 25 or so clinics in the most recent year, you can increase your achievable births by perhaps 20-30%.
I have been working on a more advanced version of the model used to produce the results above which makes more efficient use of crappy, censored data and shows results for 2021. I also plan on releasing a free app to the app store making all of this data much more usable. This post will be updated when it goes live.
Suggestion #3: Use a good PGT lab
TL;DR Make sure you use Orchid Health or Genomic Prediction for aneuploidy testing. They very likely have lower false positive and false negative rates for embryo aneuploidy when compared with other PGT labs.
It is a little known fact that there is a significant difference in aneuploidy false positive and false negative rates between PGT labs. To the best of my knowledge, there have been no randomized control trials comparing PGT labs. The best we have are independently conducted retrospective cohort analyses.
Unfortunately most of these analyses do not disclose which labs are being analyzed, making them completely useless for paients. However, I happen to know which clinics are which for one particular study submitted to ASRM in 2021. In this study, clinic A is Igenomix, clinic B is Genomic Prediction, and Clinic C is Cooper Genomics.
There’s a lot of sort of random statistics thrown out in this presentation, but I’d like to focus on those most relevant to achievable births: aneuploidy rates, pregnancy rates and miscarriage rates (which they break up into early and late miscarriages in the study)
If you watch the video, you’ll see that “Lab B (AKA Genomic Prediction)” is either as good or significantly better than the other two labs in the study across virtually every metric. If you add up the impact of these metrics, here’s what the expected number of achievable births look like for each clinic from the study:
I don’t know of any direct comparisons of Orchid Health with other PGT labs, but my guess is their false positive and false negative rates are at least low as those of Genomic Prediction. They use whole genome embryo sequencing, which means they retrieve a gigantic amount of data from each embryo. It’s likely this amount of data reduces testing errors compared to low density NGS sequencing, but I am not certain about this.
Suggestion #4: If possible, freeze embryos instead of eggs
This one is short and sweet: if you have a choice, freeze embryos instead of eggs. At a good storage facility using vitrification, about 90% of eggs will survive cryopreservation. At that same storage facility, 99% of embryos will survive. So if you already know who you want kids with, freeze embryos instead of eggs for an easy 10% boost in achievable births.
Suggestion #5: Freeze eggs or embryos as soon as possible
Since expected gain increases the more achievable births you have, all tips for maximizing it revolve around increasing the number of euploid blastocysts you can produce during IVF. You can pick a good clinic, use a good PGT lab, freeze embryos instead of eggs, and follow good prenatal care guidelines. But at the end of the day, the single biggest input variable into the “gain” equation is the age of the mother.
Here’s a graph from another research project I did showing the relationship between maternal age and number of eggs retrieved at three different clinics
You can see there’s more or less a linear decline in expected egg count per retrieval as a function of maternal age. It’s much the same story for expected zygotes and blastocysts; a linear or even exponential decline as a function of maternal age.
If you decide to do polygenic embryo screening, the sooner you start the process the better.
A compendium of other advice
When choosing a clinic, there’s several things you need to ensure they are OK with before you agree to become a patient
You need to be able to send your embryo’s biopsies to a lab of your choosing, or they need to already be working with Genomic Prediction or Orchid Health.
If you want to screen for anything other than the diseases offered by the above companies, the clinic must to be willing to implant an arbitrary embryo of your choice.
Make sure that the clinic you choose either has reasonable embryo storage costs or will let you ship your frozen embryos to a facility that does. Some clinics charge up to $1500/year for embryo storage and will raise the price on you as time goes on. Cheaper clinics charge under $1000/year for storage (some as little as $500)
Read Emily Oster’s excellent book about the things you should and shouldn’t do before, during and after pregnancy. Seriously, Oster is excellent and enjoyable to read. Ex: gardening is dangerous for pregnant women due to soil microbes but <3 drinks per week seems to be completely fine. The one possible exception to this is advice about alcohol consumption, but the science there is very complicated and confounded by selection effects.
If you’re a man over 30, consider freezing your sperm. De-novo mutations disproportionately cause conditions like cognitive deficits, severe autism and other serious conditions, and most of these mutations come from the man’s sperm.
The IVF Loss Funnel
Ok, if we put together all the data above, how many live births can you get per egg retrieval?
Naively interpreted, the above graph would imply that an average IVF cycle would only yield less than one live birth. This is true! The average IVF patient is a 36 year old woman with significant fertility issues, so it’s not particularly common for such individuals to have more than one birth per egg retrieval. In fact, over half of IVF cycles do not result in live birth. The average is dragged up somewhat by the fact that some women are able to have multiple children from a single egg retrieval.
But what if you choose a better IVF clinic and a better PGT lab than average? What if you and your partner have no known infertility issues and your female partner is younger than the average 36 year-old IVF patient (say 32 for this example).
In that case, we’d expect the graph to look something like this:
Looking better! With a younger mother, a top-tier clinic, and no history of infertility, roughly 5 achievable births per retrieval is possible.
How about in the best case scenario? Assume the following:
The mother is at peak fertility (early to mid 20s)
Egg retrieval is performed using conventional IVF hormone treatment
The father is young-ish (under 40)
Neither parent has any infertility issues
The parents use a top-tier clinic that is very good at culturing eggs into blastocysts
They use a top-tier PGT clinic with a very accurate aneuploidy test
The eggs are fertilized immediately and the resulting blastocysts were frozen
In that case, things start to look A LOT better.
The numbers above are based on a combination of sources including SART data on miscarriage and transfer loss rates, podcast episodes, publicly available data from egg donor clinics, and my own knowledge of the IVF industry. I suspect that it may be somewhat conservative for couples without infertility since I have used the infertile transfer and miscarriage rates. But they should nonetheless give a fairly accurate view of the loss funnel.
One last topic I shoud address; how much will all of this cost?
How much does IVF and PGT cost?
IVF is expensive. To do polygenic embryo screening you’ll need to pay for a consultation, ultrasounds, transportation to and from the clinic, IVF services like lab techs, medication, pre-implantation genetic testing, and data analysis services to select for non-disease traits.
I’ve called a couple of dozen top-tier IVF clinics on the phone to ask about prices. I’ll give you a general cost estimate based on those calls:
| Service | Price Range | Modal Price |
| Consultation | $50-550 | $300 |
| Follicular Ultrasound | $150-500 | $400 |
| Medication | $3000-$6000 | $4000 |
| Egg retrieval (not including transfer) | $6000-20,000 | $14,000 |
| Embryo Transfer | $3000-$6000 | $4000 |
| PGT-P | $1500-5000 | $1000 + $400/embryo |
| Selection for intelligence, height, etc | $15,000-$50,000 | $50,000* |
| Total | $9000-$88,000 | $26,500 + intelligence screening |
*I’ve heard several anecdotes of the company that offers screening for intelligence offering fairly substantial discounts to patients that are willing to provide data and testimonials, though this is not guaranteed.
This process is not cheap. If you want to do two egg retrievals and select for intelligence you’re looking at a minimum of $50k and possibly much higher depending on what you’d like to select for. I sincerely hope we can bring the price of these services down in the coming years.
TL:DR
Polygenic embryo selection can currently increase your child’s quality-adjusted life expectancy by 1-4 years, decrease their risk of various chronic diseases by 10-60%, increase their IQ by 3-10 points, increase height by up to 2.25 inches, and moderately improve other traits. The exact gain you can expect to get for each of these traits varies depending on the genetic correlation between the traits, the number of embryos you have to choose from, and the strength of the predictor used to select embryos, as well as simple luck. Subsequent children will see a somewhat smaller but still positive benefit, though for every child to benefit you will need at least 3x the number of euploid embryos as you want children.
To get these benefits, you will have to go through IVF and genetic testing of your embryos, which will cost $20k-$60k (and perhaps more depending on whether you want custom testing) and require the female partner to take 2-6 weeks off work.
You can increase the expected gain by choosing a good IVF clinic, choosing a good PGT Lab, freezing embryos instead of eggs, and beginning the process as soon as possible since younger mothers produce significantly more eggs than older mothers.
The IVF process is not particularly pleasant, and is expensive to boot, if you and your partner are willing to put up with the discomfort and expense it you can give your children advantages that are impossible to get any other way.
If you freeze embryos now, the expected gain will increase over time as the genetic predictors used to select embryos improve, and the panel of traits which you can select for will also increase.
There are technologies on the horizon that will allow for significantly greater gains across all heritable traits, making possible gains of 4 or more standard deviations across multiple traits simultaneously.
If AI doesn’t destroy the world first, the next 30 years will likely see the greatest crop of geniuses and athletes the human species has ever produced. If we are wise and select for traits like kindness, altruism, and happiness in addition to health, attractiveness and intelligence, the children born with these benefits may be able to guide the human species through the incredible upheaval and instability we are likely to see over the next century.
- How to Make Superbabies by (19 Feb 2025 20:39 UTC; 645 points)
- Significantly Enhancing Adult Intelligence With Gene Editing May Be Possible by (12 Dec 2023 18:14 UTC; 470 points)
- The Practical Guide to Superbabies by (2 Apr 2026 17:02 UTC; 210 points)
- The optimal age to freeze eggs is 19 by (8 Feb 2026 9:44 UTC; 194 points)
- Twiblings, four-parent babies and other reproductive technology by (20 May 2023 17:11 UTC; 192 points)
- Four visions of Transformative AI success by (17 Jan 2024 20:45 UTC; 113 points)
- Voting Results for the 2023 Review by (6 Feb 2025 8:00 UTC; 88 points)
- Reply to a fertility doctor concerning polygenic embryo screening by (29 May 2023 21:50 UTC; 59 points)
- Concrete positive visions for a future without AGI by (8 Nov 2023 3:12 UTC; 43 points)
- 's comment on Significantly Enhancing Adult Intelligence With Gene Editing May Be Possible by (10 Jan 2025 14:59 UTC; 37 points)
- 's comment on Why so many “racists” at Manifest? by (EA Forum; 18 Jun 2024 14:56 UTC; 31 points)
- 's comment on Superbabies: Putting The Pieces Together by (20 Jan 2026 9:56 UTC; 30 points)
- 's comment on AI Will Not Want to Self-Improve by (16 May 2023 23:30 UTC; 29 points)
- 's comment on A moral backlash against AI will probably slow down AGI development by (EA Forum; 1 Jun 2023 14:30 UTC; 8 points)
- 's comment on How to Make Superbabies by (22 Feb 2025 1:53 UTC; 6 points)
- 's comment on Cause area: Short-sleeper genes by (EA Forum; 6 Jul 2023 16:05 UTC; 4 points)
- 's comment on Are c-sections underrated? by (7 May 2023 17:08 UTC; 4 points)
- 's comment on williawa’s Shortform by (17 Apr 2026 23:14 UTC; 2 points)
- 's comment on I can see how I am Dumb by (11 Jun 2023 8:30 UTC; 1 point)

My wife completed two cycles of IVF this year, and we had the sequence data from the preimplantation genetic testing on the resulting embryos analyzed for polygenic factors by the unnamed startup mentioned in this post.
I can personally confirm that the practical advice in this post is generally excellent.
The basic IVF + testing process is pretty straightforward (if expensive), but navigating the medical bureaucracy can be a hassle once you want to do anything unusual (like using a non-default PGT provider), and many clinics aren’t going to help you with anything to do with polygenic screening, even if they are open to it in principle. So knowing exactly what you want and what you need to ask for is key.
Since this post was written, there have been lots of other developments and related posts in this general area:
Significantly Enhancing Adult Intelligence With Gene Editing May Be Possible
Superbabies: Putting The Pieces Together
Gameto Announces World’s First Live Birth Using Fertilo Procedure that Matures Eggs Outside the Body
Overview of strong human intelligence amplification methods: Genomic approaches
And probably many others I am forgetting. But if you’re a prospective parent looking for practical advice on how to navigate the IVF process and take advantage of the latest in genetic screening technology, this post is still the best place to start that I know of. Some of the things in the list above are more speculative, but the technology for selection is basically ready and practical now, and the effect size doesn’t have to be very large for it to beat the status quo of having an embryologist eyeball it.
I think this post is a slam dunk for a +9 and a spot in the LW canon, both for its object-level information and its exemplary embodiment of the virtue of empiricism and instrumental rationality. The rest of this review details my own experience with IVF in the U.S. in 2024.
This section of the original post basically covers it, but to recap, the two main things you’ll want to ask your prospective IVF clinic are:
Can we use Orchid Labs or Genomic Prediction for PGT?
Can we implant any healthy embryo of our choosing? (some clinics can have policies against sex selection, etc.)
In my experience, the best time to ask these questions is in-person at your initial consultation; it can be hard to get answers over the phone / in email before you’re at least a prospective patient, since they generally require a doctor or NP to answer.
The good news is, if you get affirmative answers to these questions, you mostly don’t need to worry about whether the clinic or your embryologist is skeptical or even outright hostile to polygenic screening, because you can simply request your sequence data from your PGT provider directly and have it analyzed on your own.
Note: Genomic Prediction offers their own polygenic screening test (LifeView), but if you’re planning to have a third party analyze the sequence data for non-disease traits, you don’t need this. You can just have your IVF clinic order PGT-A tests from GP, and then request your raw sequence data from GP directly once you get the PGT-A results. AFAIK the actual sequencing that GP does is the same regardless of what test you order from them, and they’re happy to share the raw data with you if you ask.
Another thing you’ll want to confirm is whether you can store any aneuploid embryo(s) long-term. Aneuploid embryos are typically considered not viable and most clinics won’t even try to implant them. But they’re worth keeping frozen speculatively, in case future technology allows them to be repaired, etc. Some clinics will have a policy of automatically discarding confirmed-aneuploid embryos unless you make specific arrangements to do something else with them. Usually this will be a question in a big packet of paperwork you’ll have to fill out about what you want to do with the embryos / eggs in various scenarios, e.g. death / divorce / time limit etc. so just make sure to read carefully.
On selecting a good-quality IVF clinic: the live birth metrics in this post are a good starting point, but probably confounded somewhat by the population the clinic serves, and realistically the biggest factor in determining how many embryos you get is going to be your personal health factors and age. My wife is over 30 and in pretty good shape and took a bunch of vitamins before / during the cycles (B12, Omega-3, Myo-Inositol, a prenatal vitamin) and we ended up with 21 eggs retrieved across two cycles, which is right around the expected number for her age.
These attrited down to 10 mature embryos during the fertilization process, 5 of which were screened out as aneuploid via ordinary PGT-A. We had the remaining 5 embryos polygenically screened.
We’re planning to start implanting next year, so I can’t speak to that part of the process personally yet, but overall we’re very happy with the results so far. There was a clear “winner” among the embryos we screened that will be our first choice for implantation, but it’s nice to have all the data we can on all the embryos, and depending on how things go we may end up using more than one of them down the line.
The polygenic screening wasn’t cheap, and given the number of embryos we had to select from, the maximum possible benefit is pretty mild (2.3 bits of selection if we only use one of the 5 and it successfully implants). But given the hassle and expense of IVF itself (not to mention pregnancy and raising a child...) it seems overwhelmingly worth it. We were considering IVF for fertility preservation reasons anyway, so the main question for us was the marginal cost of the extra screening on top.
I’d like to write a longer post with my own takes about having / raising kids on the eve of AI, but for now I’ll just say a few things:
The choice about whether to have kids is always a personal one, but right now seems like as good a time as any, historically speaking, and takes like these seem crazy wrong to me. Even if you’re thinking purely in terms of the advantage you can give your kids (probably not a good idea to think this way), by far the biggest advantage is being marginally earlier. If you’re interested in having kids but worried about the costs or effectiveness of current IVF / screening methods, consider just having kids the old-fashioned way instead of delaying.
I have short timelines and expect the default outcome from ASI being developed is swift human extinction, but I still think it’s worth having kids now, at least for me personally. My wife and I had happy childhoods and would have enjoyed being alive even it was only for a short time, and hopefully that’s at least a somewhat heritable trait. And regardless of the endpoint, I expect things to be pretty OK (even great) at least for me and my family, right up until the end, whenever that is. Despite being a “doomer”, I am long the market, and expect to be pretty well-off even if those particular bets don’t pay off and the larger world is somewhat chaotic in the short term.
2.3 bits of selection on a single kid realistically isn’t going to make a difference in any kind of “Manhattan project for human intelligence enhancement” and that’s not why we did it. But my sense from having gone through this process is that the barriers to some of the things that Tsvi describes here are more social and financial and scale than technical.